Historical Plaster and the Irreplaceable Lime
The Lime of Our Lives


Lime may very well be the most prized and versatile building material of the civilized world. The Mayans, Egyptians, Greeks and Romans all used lime extensively. The fact that many of their works realized in lime have survived hundreds or even thousands of years until the present day testifies to the durability of lime. The benefits of being physically durable seem obvious enough; however, beauty is an oft overlooked factor in sustainability. After all we’re often willing to go to great lengths preserve and maintain what aesthetically pleases us and are likewise eager to tear down what we find ugly.
Leonardo da Vinci, Michelangelo and Raphael are considered the three preeminent masters of the Renaissance, each producing lime plastered masterpieces delicately painted al fresco. Lime has likewise been modified to construct sublime, enduring structures such as the 2nd century Roman Pantheon as well as our own Hoover Dam of the 20th century.
Although lime can be produced from a variety of natural sources such as marble and shells, lime manufactured for plaster typically comes from limestone. Limestone is a sedimentary rock that forms from the skeletal remains of marine creatures that have accumulated on the ocean floor. With time and pressure these remains are pressed together into beds of stone. The Garden of the Gods in the Shawnee National Forest provides one stunning example of exposed sedimentary limestone.
Chemistry & Manufacture
Many marine organisms have the ability to metabolize calcium (and occasionally magnesium) ions present in the water to form shells or skeletons for themselves. Actually, we share this ability and use it to grow our own skeletons and teeth. Pure limestone is a carbonate of calcium or “calcite” having the formula CaCO3. Limestones that have significant percentages of magnesium are referred to as dolomitic limestones. Both are useful for building as well as manufacturing lime for plasters and mortars.
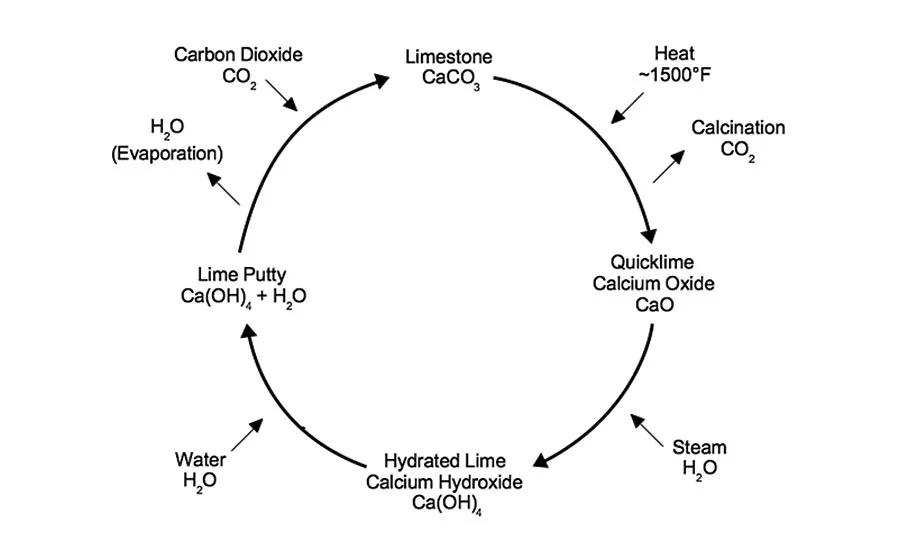
Like gypsum rock, limestone must be baked in preparation for its use as a plaster. Unlike gypsum however, this occurs at a higher temperature that makes it a moderately energy intensive process. Limestone can be efficiently baked at temperatures starting at 1,500 degrees F. At this temperature lime “calcines” or off-gases carbon dioxide, CO2. The resulting material has the chemical formula calcium oxide or CaO, commonly known as “quicklime.” Although quicklime has and can be used as a plaster, it is extremely alkali and exothermically reactive even explosive under certain conditions. Unless engaged in very specific restoration work most plasterers will utilize lime that has undergone further processing.
By adding water as a fine mist or steam the reactive quicklime can be “quenched,” converted to a relatively inert dry hydrated form with the chemical formula calcium hydroxide or Ca(OH)2. This process is known as “slaking” and the resulting powdery binder is commonly called a dry hydrate or slaked lime. If excess water is added a lime putty will form. So long as the putty is protected from exposure to air it will last indefinitely.
Generally, the longer a putty sits the creamier and more desirable it is for plastering. Both the dry hydrate and putty forms of lime have a lower alkalinity than quicklime making them the considerably safer preference for plasterers to work with. When exposed to air under normal conditions the processed lime slowly returns to its natural state of calcite, resembling the limestone from which it was derived and recapturing from the air much of the CO2 lost in manufacturing through a process known as “carbonation.”
Properties & Specifications
Lime has been used for just about everything: interior plaster, exterior stucco, mortar for bricks and stone, mouldings and ornament. The list of positive qualities of lime is extensive. First of all, limestone is a plentiful resource found in large quantities distributed all over the world. Although the burning of limestone requires far more energy than gypsum, limestone can still be baked at temperatures low enough to utilize renewable, natural materials that require a fraction of the production energy of modern materials, such as Portland cement or polymers. Furthermore, limes have the ability to re-absorb the CO2 lost in manufacturing through the process of carbonation, illustrated by the famous lime cycle.
Lime plasters are incombustible, breathable and maintain a low pH or alkalinity that actively suppresses mould growth and promotes healthy indoor air quality. Limes are chemically compatible with all other heritage binders; blends with clay, gypsum, hydraulic limes and natural cements have numerous practical applications. Lime is not only very white but also very bright, that is to say it has a high index of refraction of visible light so that the addition of mineral tints can achieve an extensive range of saturated colors.
Not only do lime plasters maintain a good surface hardness and erosion resistance making them ideal as an exterior stucco, they have a high tensile strength conferring the quality of flexibility. This means they can literally bend over time with less propensity of breaking or spalling. In conjunction with this flexibility lime has the property of autogenous healing. Because there is always a percentage of “free” or uncarbonated hydroxide in the plaster, should a crack occur lime literally has the ability to crystalize or knit itself back together.
Despite all of the deserved praise there are a few limitations and precautions to consider when working with or specifying limes for plaster. Lime is not self-binding. Careful attention has to be given to the distribution of aggregates and fibers to make a high performance lime plaster. Closely related to this attribute is that lime shrinks upon drying. This place a limit how thick lime plasters can be applied and therefore how quickly work can proceed. As all of the water eventually evaporates from the system, lime plasters must be compressed and burnished while they are drying so that they don’t end up being friable, subject to crazing (microcracks) or even delamination.
Carbonation, the chemical cure of the plaster needs to occur for every layer and takes time, sometimes days or even weeks to complete. This often requires interim protection of the plaster surface from the sun, wind and limiting exterior work to times of year where the lime plaster will not be subject to freeze thaw cycles. In summary, lime is a true craft material that requires a good deal of mixing and application knowledge on the part of the plasterer.
Some limestones are “contaminated” by materials that drastically affect the chemical properties of the resulting lime during the baking process. In our next essay we’ll begin taking a closer examination of another member of the lime family, the hydraulic limes.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!






